Ethanol
| It matters Chemistry |
| Action and reaction |
| Elementary! |
| Spooky scary chemicals |
| Er, who's got the pox? |
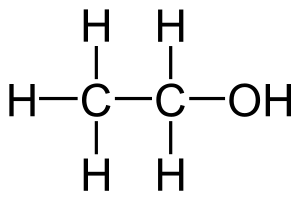
Ethanol is a chemical substance with the formula CH3CH2OH. That is, a two carbon alkyl chain (ethan-) attached to an alcohol functional group (-ol). Ethanol can be synthetically produced (i.e.: from fossil petrochemicals, usually via catalyzed hydration) or produced via biological fermentation.
Drinkies?[edit]
- Main article: Alcohol
It is the primary alcohol found in consumable drinks and in the bloodstreams of RationalWiki editors. While in the body, it oxidises to ethanal (acetaldehyde) which is one of the causes of a hangover and eventually to ethanoic acid (acetic acid, the reason that bad wine tastes of vinegar). A related homologous compound, methanol (CH3OH) is in fact a poison, being able to pass through the skin and eventually cause blindness and death. Ethan-diol, (HO-CH2CH2-OH) is another related compound that is used as antifreeze, like methanol, and is also used to spike really shit drinks and could be lethal. Ethanol, however, is "mostly harmless" unless taken in much slightly larger quantities.
Fuel[edit]
Bio-fuel[edit]
Ethanol can be used as a bio-fuel. Mostly it is formed from corn crops and is advertised as an environmentally friendly way of producing fuel. In theory it is a carbon-neutral way of producing energy-dense fuel from environmental nutrients, but in fact the industrial-scale agriculture required to produce meaningful amounts relies almost completely on fossil fuels, to the extent that more petroleum is required to produce a given amount of bio-ethanol than would be needed to simply refine an equivalent quantity of gasoline. Even ignoring this, the production of bio-ethanol competes with other agricultural products and with land conservation. For these reasons among others, it has recently been viewed as a bad thing rather than the solution to the pending energy crisis. In principal, production of ethanol from fermenting waste biomatter unusable for any better purpose isn't damaging to the environment or society.
Petrol additive/substitute[edit]
Ethanol can be added to petrol/gasoline to reduce fossil fuel use, or it can replace petrol entirely. Ethanol-gasoline mixtures are usually labelled by the percentage of ethanol they contain; for example, E10 is 10% ethanol and 90% gasoline, while E85 is 85% ethanol and 15% gasoline. (This has nothing to do with the E numbers used in Europe for food additives.) In some countries, such as Brazil, all 'gas' contains a large amount of ethanol, while ethanol is also sold in a nearer-100% concentration.
This has both advantages and disadvantages. As noted above, ethanol is much better CO2 emissions-wise than straight petrol. Ethanol also burns hotter and more efficiently, but only in vehicles designed for it, those not specifically designed for it will work worse or be damaged. Nevertheless, unless the ethanol is produced from waste produce it can have a negative impact on food production. Also, the use of ethanol in cars produces significantly more Ozone (O3) - a harmful pollutant at ground-level - than petrol does. There is also the fact that ethanol only has about 2/3 the energy density of pure gasoline, which significantly reduces mileage, even in low-concentration blends, such as E10.
Rocket fuel[edit]
In the 1940s it was used as a successful rocket fuel to cope with wartime seizures and blockades of oil fields. The German V2 rockets were powered by a mix of ethanol and water not too dissimilar to Bacardi 151 and Stroh 80 - the former comes equipped with a flame arrestor on the bottle to prevent it going up like a bottle of ether.